van Ingen lab and collaborators discover a new way to assemble chromatin
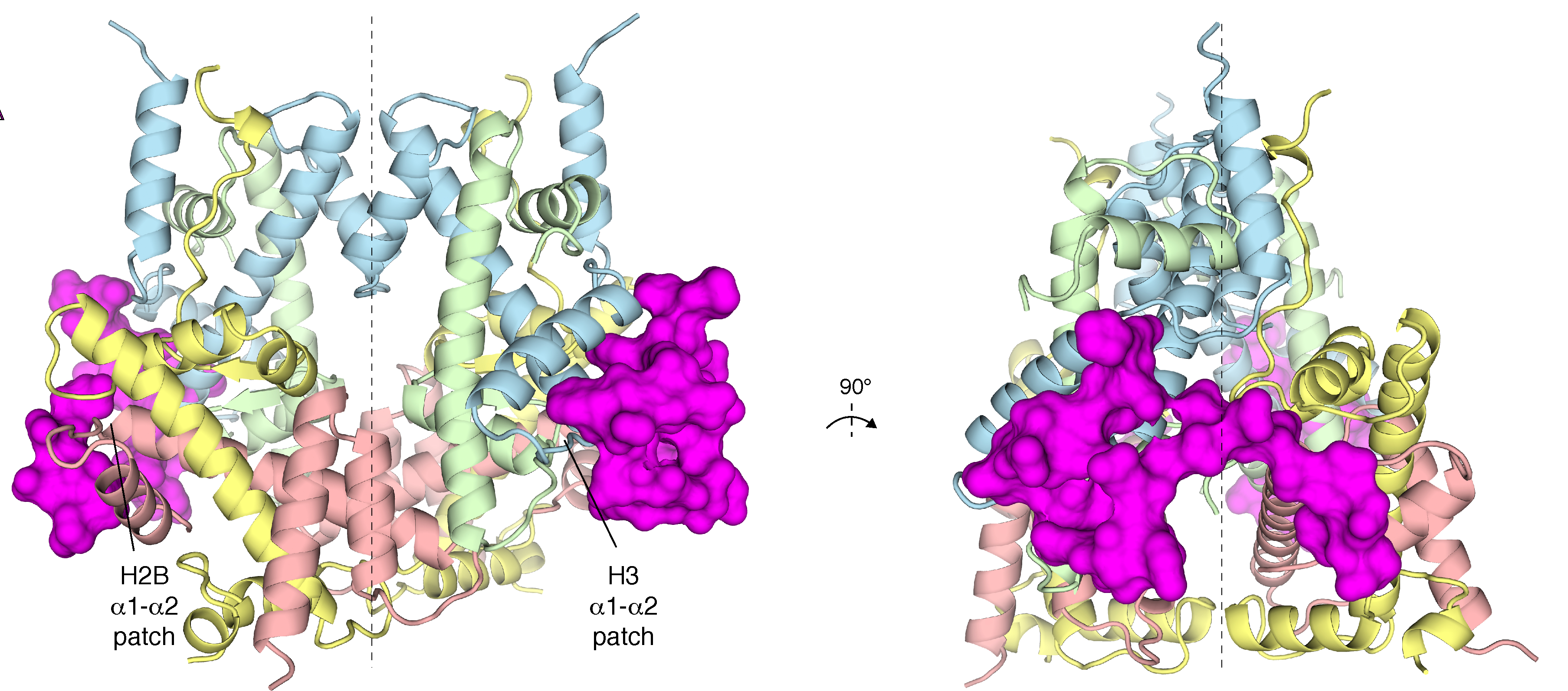
Image: The tructure of APLF (magenta), bound to the histone complex.
A team of researchers led by the group of Hugo van Ingen, discovered a novel mechanism by which nucleosomes, the building blocks of chromatin, can be assembled. Nucleosomes are made by carefully controlled process in which different histone proteins are deposited step-by-step on the DNA through different chaperone proteins.
The chaperone protein APLF can assemble the nucleosome single-handedly in one step, the researchers write in the scientif journal Science Advances. As the APLF protein is part of the DNA repair process, this unique mechanism could be means to maintain epigenetic information during DNA repair.
PhD student Ivan Corbeski in the van Ingen group used NMR and other experiment to discover that APLF can bind and assemble all four core histone proteins simultaneously,. Together with the Sixma group at the Netherlands Cancer Institute NKI the crystal structure of the APLF-histone complex could be solved, showing that APLF assembles the histones in their native fold as in the nucleosome.
The group of Francesca Mattiroli at teh Hubrecht Institute could then show that APLF can transfer the histones via this complex to the DNA to form nucleosomes. These findings were then confirmed in cellular experiments in the group of Haico van Attikum at Leiden University Medical Centre.
Read more about research in the lab of Hugo van Ingen
Publication:
Chaperoning of the histone octamer by the acidic domain of DNA repair factor APLF
Ivan Corbeski, Xiaohu Guo, Bruna V. Eckhardt, Domenico Fasci, Wouter Wiegant, Melissa A. Graewert, Kees Vreeken, Hans Wienk, Dmitri I. Svergun, Albert J. R. Heck, Haico van Attikum, Rolf Boelens, Titia K. Sixma, Francesca Mattiroli, Hugo van Ingen
SCIENCE ADVANCES, 27 Jul 2022, Vol 8, Issue 30
DOI: 10.1126/sciadv.abo0517
